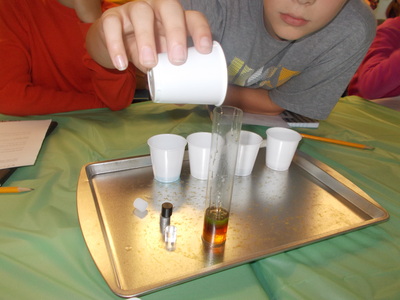
The students experimented with the various densities of liquids during class today. They were given five liquids: water (tinted blue), dish soap (green), honey (tan), syrup (dark brown), and vegetable oil (light yellow). They first predicted the order, then we poured each into a cylinder to observe what happened. We recorded the typical density of each substance. Next, we added four different cylinders: aluminum, acrylic, polyethylene (the white cylinder), and acetal delrin (the black cylinder). They were able to see that the aluminum cylinder sank to the bottom since it has a higher density than honey. Next, the black cylinder stopped between the honey and syrup, the clear cylinder stayed between the syrup and dish soap, and the white cylinder stayed between the water and oil. It was interesting for the students to see solids that would float in some materials, but sink in others. We were able to relate this to math (decimal place value- including comparing and ordering) by figuring out what each solid cylinder's density must be close to.
Afterwards, we added isopropyl alcohol (rubbing alcohol) that was dyed red. It was the least dense and stayed on top of the oil. It also smelled the worst- an unwelcome surprise for those who thought it was Kool-Aid!
Afterwards, we added isopropyl alcohol (rubbing alcohol) that was dyed red. It was the least dense and stayed on top of the oil. It also smelled the worst- an unwelcome surprise for those who thought it was Kool-Aid!